Our chemical research focuses on improving the sustainability of synthetic polymers, either by designing them for degradation or developing route to repurpose them post-use. We are also interested in renewable energy and energy storage. For more details, click on the links below!
Chemical Research
Targeting more sustainable organic materials and their applications Visit Archived Chemical ProjectsMeasuring, Modeling, and Mapping Microplastics in the Atmosphere of Michigan
Repurposing Plastics for a More Sustainable Future
Synthetic polymers have an enormous impact on our lives, yet the manner in which they are produced, used, and disposed of is unsustainable. Globally, we produce over 300 million tons of plastics per year, and a stunning >90% of plastics are made from petroleum feedstocks and only a scant 9% of plastics are recycled. Products that are recycled through current mechanical processes are frequently downgraded into lower-quality materials. We are currently exploring methods for “chemical recycling”. This approach includes developing depolymerization procedures and synthetic methods for repurposing degraded polymers into equal-quality or value-added materials.
Listen to Anne’s interview on NPR’s Stateside where she talked about recycling plastics.
Coates, G. W.; Korley, L. T. J.; McNeil, A. J. Challenges and Opportunities in Sustainable Polymers. (editorial) Acc. Chem. Res. 2022, 55, 2543–2544.
Jha, R. K.; Neyhouse, B. J.; Young, M. S.; Fagnani, D. E.; McNeil, A. J. Revisiting poly(vinyl chloride) reactivity in the context of chemical recycling. Chem. Sci. 2024, 15, 5802–5813.
Fagnani, D. E.; Kim, D.; Camarero, S. I.; Alfaro, J. F.; McNeil, A. J. Using Waste Poly(vinyl chloride) to Synthesize Chloroarenes by Plasticizer-mediated Electro(de)chlorination. Nature Chem. 2022, 15, 222–229.
To read the article, click here.
- Highlighted by Helms in Nature Synthesis
- Highlighted by Chemistry World.
- Listen to this interview on NPR Stateside!
- Highlighted in the UM Record.
- Highlighted by UM News.
- Covered by the local CBS News!
- Highlighted by ClickonDetroit.
- Highlighted by The Optimist Daily.
- Highlighted by ScienceDaily.
- Highlighted in MaterialsToday.
Redox-active Molecules for Energy Storage
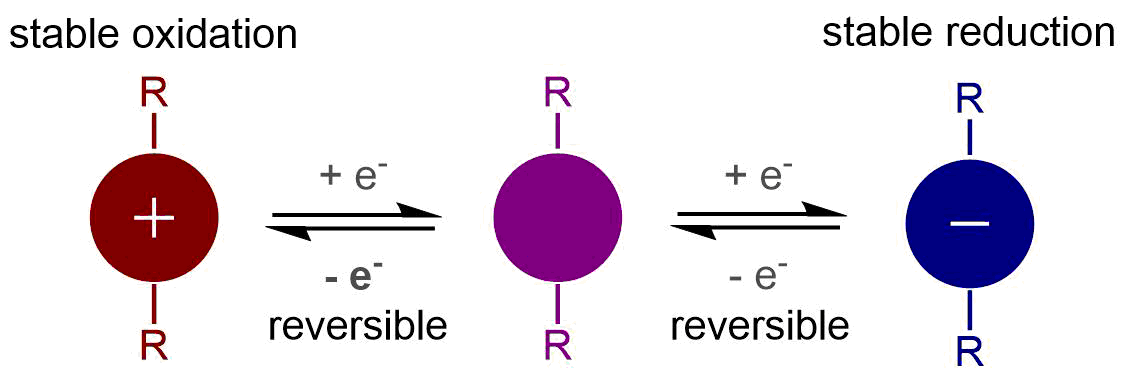
As renewable, but intermittent energy sources become more prevalent, there is an increasing need for scalable, high-capacity methods to store energy. One of our group’s newer projects looks at developing soluble, high-potential organic redox active materials for energy storage applications such as non-aqueous redox flow batteries. We are especially interested in “ambipolar” molecules which can be both oxidized and reduced, enabling devices with the same active species on both sides of the battery. Using a combination of synthetic and electrochemical methods, we are exploring the relationships between molecular structure and properties including potential, cycling stability, and solubility. This work is part of a large, collaborative grant – JCESR – funded by the DOE.
Singh, S.; Tami, J. L.; Gruich, C.; Gatz, A. J.; Dong, J. Nguyen, B. H.; Smith, J. A.; Goldsmith, B. R.; McNeil, A. J.; Kwabi, D. G. Sulfonated Benzo[c]cinnolines for Alkaline Redox-Flow Batteries. ACS Appl. Energy Mater. 2025, ASAP.
Asserghine, A.; Kim, S.; Vaid, T. P.; Santiago-Carboney, A.; McNeil, A. J.; Rodríguez-López, J. Ionic Strength Impacts Charge Capacity in a Redox-Matched Flow Battery: From Single-Particle Interrogation to Battery Cycling. ACS Energy Lett. 2024, 9, 2826–2831.
Tami, J. L.; Mazumder, M. M. D.; Cook, G. E.; Minteer, S. D.; McNeil, A. J. Protocol for Evaluating Anion Exchange Membranes for Nonaqueous Redox Flow Batteries. ACS Appl. Mater. Interfaces 2024, 16, 53643–53651.
Microplastics Capture & Repurposing
The spread of microplastics throughout the environment is a growing problem with potential for long-term harm to the nation’s ecosystems. Numerous studies have quantified their presence, identified their sources, and pointed out their negative impact on human and environmental health. One major source of microplastics is wastewater, for example effluent from washing machines that contains fibers from clothing. While many larger plastic particles can be sequestered by municipal wastewater treatment facilities, a sizable number of smaller plastics still convey. This project will develop efficient microplastic remediation strategies to capture and eliminate this source of pollution before it enters our waterways. In addition to this goal, the materials used to capture the microplastics will themselves be made of recycled plastics, and the captured microplastics will be upcycled into industrially usable materials. Together, these three project goals will bolster a sustainable plastics economy with less reliance on fossil fuels as well as reduced plastic waste in rivers, lakes, and oceans. This project is part of a funded EFRI through the NSF, and is in collaboration with colleagues Prof. Jose Alfaro, Prof. Brian Love, and Prof. Paul Zimmerman.
See this press release from UM about the grant for more information.
See this highlight of Anne’s public lecture (Nov 18) on microplastics pollution and solutions.
Ramkumar, M.; Ji, W.; Thurber, H. E.; Clough, M. E.; Chirdon, S.; McNeil, A. J. Enhancing microplastic capture efficiencies with adhesive coatings on stainless-steel filters. RSC Appl. Polym. 2024, 2, 456–460.
Milne, S. L.; Ramkumar, M.; Rieland, J. M.; Thurber, H. E.; Love, B. J.; McNeil, A. J. Informing the Public about Microplastics through a University and Museum Partnership. J. Chem. Educ. 2024, 101, 97–103.
Chazovachii, P. T.; Rieland, J. M.; Sheffey, V. V.; Jugovic, T. M. E.; Zimmerman, P. M.; Eniola-Adefeso, O.; Love, B. J.; McNeil, A. J. Using Adhesives to Capture Microplastics from Water. ACS ES&T Engg. 2021, 1, 1698–1704.
PFAS Remediation of Contaminated Groundwater
Per- and polyfluoroalkyl substances are both pervasive and persistent. Dubbed ‘forever chemicals’ due to their resistance to biodegradation, a staggering ~98% of the US population has measurable levels of PFAS in their blood. Remediation strategies are urgently needed to attenuate the detrimental impacts of PFASs. One of the most promising remediation technologies involves adsorption of PFASs onto substrates. Our group is pioneering a new class of adsorption materials for PFAS remediation.

